How to use super-resolution microscopy for detailed investigation of functional nanoparticles
SuperCol is all about developing highly functional colloidal particles for biomedical applications. To do this in a clever, strategic way, it is crucial to know exactly what the synthesized particles look like. How many functional molecules are present at their surface? How are those distributed? It seems obvious to establish such important features using the imaging power of super-resolution microscopy. However, this requires a dedicated approach where a few troublesome issues have to be solved. This is why super-resolution microscopy is at the heart of the first work package of the SuperCol project.
There’s a bright outlook for the functionalised colloidal particles that are being developed within SuperCol. Think of super-selective biosensors for Covid19 and cholera, responsive particles that capture and quantify inflammation proteins. To arrive at such applications, SuperCol researchers develop methods to equip the particles with functional bio-active molecules. These enable the particles to interact at a cellular level, for instance binding to pathogens and biomarkers, or detecting membrane receptors.
“Exactly how this interaction develops determines their performance as, for instance, a biosensor”, explains SuperCol coordinator Dr Peter Zijlstra. “And since this all depends on the biomolecules we attach to their surface, you can imagine that we need to know all about that. How many of the molecules actually end up at the surface of the particles? Are they neatly distributed, are they in clusters, or maybe just on one side of the particle? And even more in detail: do they retain their structure, or do they fold up and loose functionality?”
Super-resolution microscopy to the rescue
The answers to Zijlstra’s questions are hard to get because the particles are extremely small: roughly a thousand times smaller than the width of a human hair. This implies that they can’t be studied with a common optical microscope. It is fundamentally impossible to resolve details that are smaller than half the wavelength of the light being used. For visible light, this diffraction limit lies between 200 and 750 nanometers (nm), depending on the colour of the light. It means that most nanoparticles (sizes 100 nm or smaller), many proteins (around 10 nm) and all simple molecules (1 nm) cannot be adequately imaged.
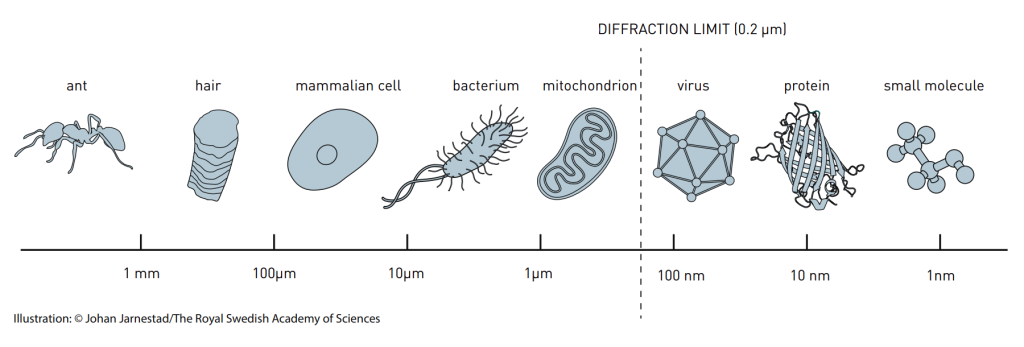
Of course, using even shorter wavelengths could solve this. However, such UV and X-ray systems are expensive, often suffer from a lack of contrast and may damage the sample. Here, super-resolution microscopy comes to the rescue. In 2014, the Nobel Prize in Chemistry was awarded to Eric Betzig, W.E. Moerner and Stefan Hell for the development of an approach that ‘brings optical microscopy into the nano dimension’. The basic principle underlying this ‘breaking of the diffraction limit’ is the use of blinking fluorophores. These are small molecules that are capable of fluorescence: they can emit light themselves. Capturing the light of these tiny emitters from within the sample and imaging it on a camera allows you to pinpoint the exact location of the fluorophore with a precision that is more than ten times better than the diffraction limit. As a result, much smaller details can be imaged that have remained invisible with regular optical imaging. Of course this is easier said than done and there’s a multitude of super-resolution techniques to make the concept work. But in the field of biology it has become quite common to image details of only a few tens of nanometers.
The mislocalisation problem
It now seems clear how to study the molecular details of the SuperCol particles: just attach fluorophores to the bio-active surface molecules and use super-resolution microscopy to determine their location. Alas, there’s a catch here. As Dr Rodolphe Marie explains, super-resolution microscopy is mostly used with biological samples, where it can for instance reveal the molecular details of cell membranes. “One of the reasons it works there is that the materials of biology themselves show no interaction to super-resolution microscopy. They are transparent both to the signals of the fluorophores and to the laser light that is being used to induce the fluorescence. With our nanoparticles that is quite different.”
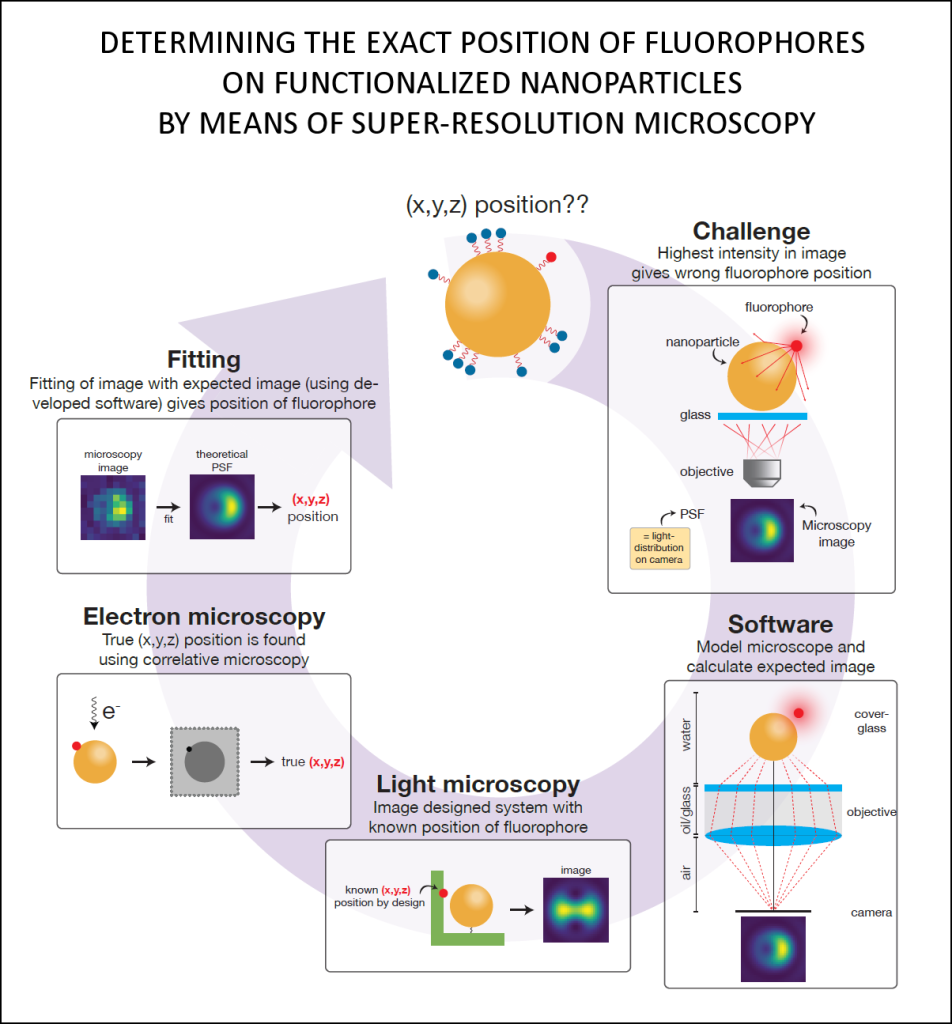
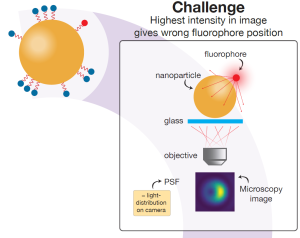
According to Marie, principal investigator in work package 1, the particles often have a high refractive index, their curved surface acts as a lens, and so-called plasmons can occur, which are interfering optical resonance phenomena. “As a result, when you apply super-resolution microscopy you not just collect light from fluorophores, you collect light from the particle-fluorophore system.” This, Marie explains, poses a major concern. Because the fluorescence signal is not collected in a straightforward manner, the super-resolution microscopy image might misrepresent the actual location of the fluorophore. This mislocalisation of course severely undermines the research purpose of obtaining insight into the precise location and distribution of molecules at the surface of a nanoparticle.
Correlative approach
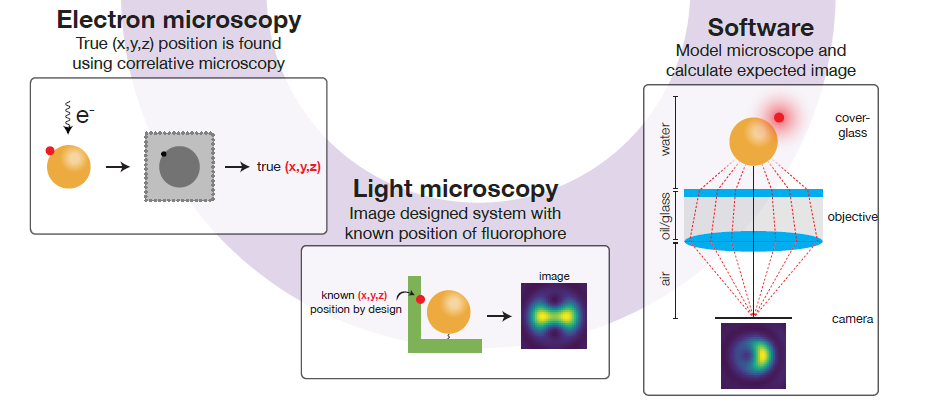
So what to do about this? In this work package, the SuperCol team aims to develop software that can correct for the mislocalisation phenomenon. To be able to do this, the research follows a correlative approach where super-resolution microscopy is applied in tandem with electron microscopy. The latter is used to establish the true position of the fluorophore so that comparison of the two methods will provide data on the mislocalisation. This will then be used to validate the software.
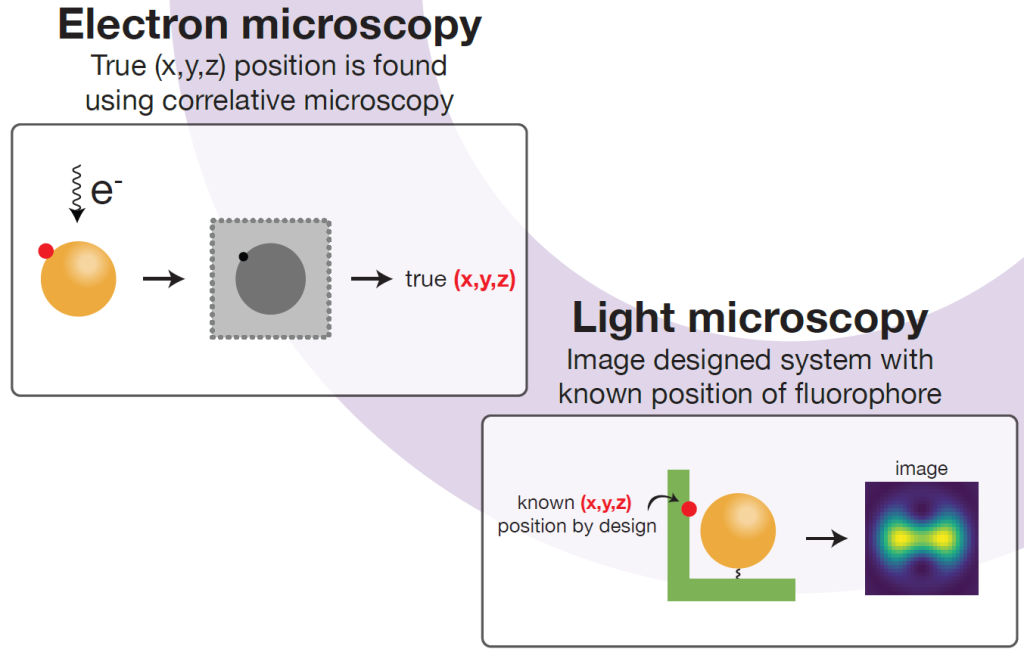
SuperCol PhD candidate Iain Harley will use cryo-electron microscopy to benchmark the super-resolution microscopy for localization of single molecules. According to Marie, developing the electron microscope for imaging the nanoparticles is in itself quite a big undertaking. “It’s not just benchmarking, it is really establishing how to use electron microscopy in a way that enables the comparison with optical super-resolution microscopy.”
Zijlstra adds that this indeed requires the dedicated effort of a specialized PhD candidate. “And we take advantage of the new electron microscope that MPI-P has installed in 2021. Actually, that microscope is more expensive than our whole project!” Harley will be investigating samples delivered by the other SuperCol PhD candidates. In particular Masih Fahim, Sarojini Mahajan, Bahar Rouhvand and Aleksandra Adamczyk will contribute to the study.
Corrective software
The experimental results will enable PhD candidate Teun Huijben to validate the software he is developing to correct for the mislocalisation in a super-resolution image. He recently achieved a milestone with an application that predicts the super-resolution image of fluorophores near spherical nanoparticles. The software models the so-called point spread function (PSF), which basically describes how a tiny fluorophore light source is imaged by the microscope camera as a somewhat fuzzy ‘blob’- quite similar to the image of a star seen through a telescope.
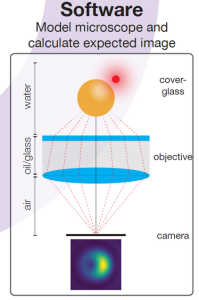
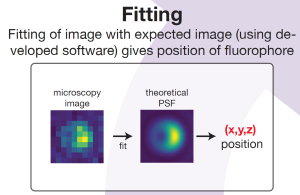
Based on an analytical representation of the light path in the microscope, Huijben’s software provides an exact PSF calculation for fluorophores near a nanoparticle and is three orders of magnitude faster than available approaches. It will be published as free open-source software while commercial numerical software packages require an expensive license.
With respect to the SuperCol research it is relevant that the software is very fast and quite versatile. “The nanoparticle can be of any size and material”, Huijben explains, “and the fluorophore can have any position and orientation with respect to the nanoparticle. This makes it a powerful tool to investigate the mislocalisation issue and ultimately provide a method to correct for it.”
A broad range of nanoparticles
All in all, the SuperCol researchers in this work package will investigate a broad range of particles including silica, silicon, polymers and metallic nanoparticles. These are synthesised in different shapes and sizes to obtain a comprehensive understanding of the impact of particle geometry and optical properties on the localisation accuracy. Marie: “We want a complete view of the physics, so we include multiple materials. But it’s also from the perspective of application. The focus of SuperCol is on biosensing, but functionalised colloidal particles are relevant to many other applications. Think of targeted drug release systems, or fabricating nanostructures.”
Zijlstra adds that the extension of the super-resolution technique to other material systems beyond biology is in fact one of the driving forces for the project as a whole. “We take it from imaging of biological nanostructures to the broader field of imaging of solid-state nanomaterials. This is also reflected by partners of the project that are outside the fields of bio-imaging or medicine. There’s a broad interest in doing super-resolution microscopy beyond the biological domain, and the SuperCol research will surely contribute to evolving the technique towards imaging all kinds of materials at the nanoscale.”